Closed System Manufacturing for Cell Therapy Development
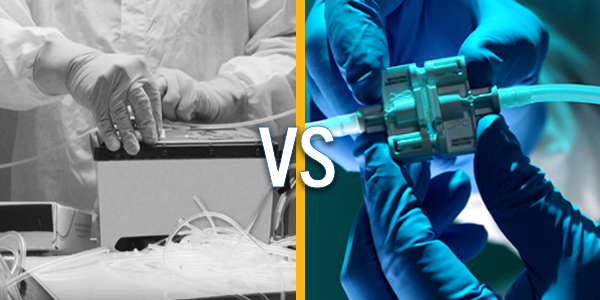
The promise of CGT is breakthrough and the people who are rapidly evolving this space are doing extraordinary work. But, the cutting-edge work is ahead of the process – the integrity of the process does not match the integrity of the product. Tube welding equipment is a burden and introduces more risk to the process. It takes a skilled operator 4X as long compared to using sterile connectors, is a larger capital investment, introduces risk (of downtime, operator error, contaminants, etc), and requires continuous maintenance and validation.
With an increased focus on efficiency and flexibility in cell therapy development accelerated the adoption of a closed system design utilizing single-use technology (SUT). Single-use connectors offer an easy-to-use method for maintaining flow path sterility and integrity while enabling the protection needed to avoid costly failures from contamination.

Cryopreservation in cell therapy and gene therapy (CGT) manufacturing is critical to maintaining product integrity. Learn how to improve aseptic processing; considerations and what to look from components used in cryogenic freezing of cells.

Cells in small-volume Advanced Therapy Medicinal Products (ATMP) typically are preserved at ultra-low temperatures. Learn about cryopreservation challenges, best practices and the latest advances in single-use products, including the new MicroCNX® Nano Series connectors, which are made to meet the rigorous demands of today’s CGT processes.

Small-batch aseptic processing has become integral to producing these precision medicine treatments. This shift, coupled with downstream filtration challenges and a widespread push to close aseptic processes earlier in upstream development, has driven manufacturers to reevaluate how to optimize sterility throughout the process.

Is your manufacturing process still tied up using tube welding? Have you considered the efficiencies that could be achieved with aseptic connectors? Compare the differences that may help you determine if your process could be enhanced by aseptic connectors.
CPC's Biopharmaceutical Product Catalog provides specifications on CPC connectors purpose-built for bioprocessing, cell therapy, and gene therapy applications.
