PAT: Build Quality into Biopharmaceutical Processes with Real-Time Monitoring and Control
Process Analytical Technology (PAT) ensures quality in biopharmaceutical manufacturing by monitoring and controlling processes in real time. It utilizes analytical tools to develop manufacturing processes that accommodate material and equipment variability. Once critical process parameters (CPPs) impacting critical quality attributes (CQAs) are identified, analytical methods are employed to monitor and control CPPs, maintaining them within the desired design space. This approach integrates quality by design (QbD) principles into the process rather than relying on product testing only in the end.
Download the assets below to learn more on the solutions available to implement PAT into your bioprocess.
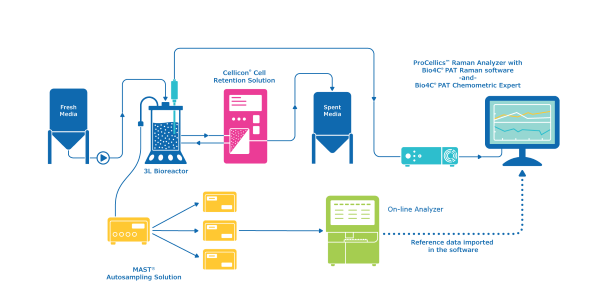
This application note describes use of the ProCellics™ Raman Analyzer with Bio4C® PAT Raman Software (also known as Raman PAT Platform) to perform inline and real-time measurement of TCD, VCD and the concentration of glucose, lactate and ammonium in a bench-scale bioreactor

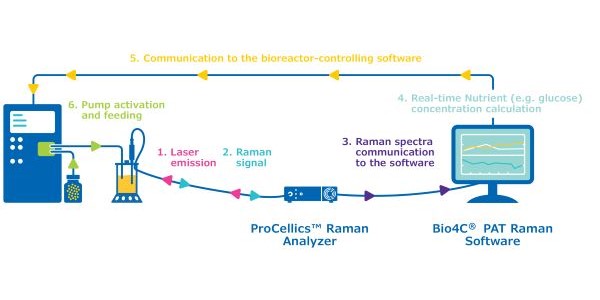
This application note describes the use of ProCellics™ Raman Analyzer with Bio4C® PAT Raman Software to monitor glucose levels in a bioreactor and trigger automated addition of feed to maintain the desired concentration
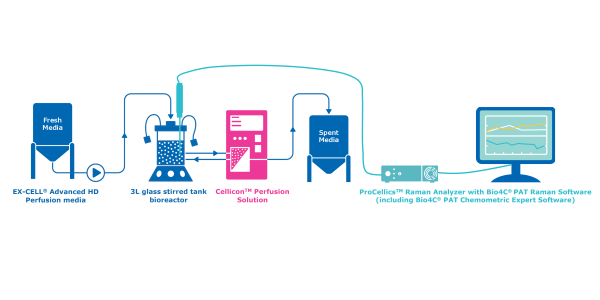
This application note introduces a case study for the implementation of a Raman spectroscopy soft-sensor for in-line and real-time monitoring of critical process parameters (CPP) in mammalian perfusion cell cultures
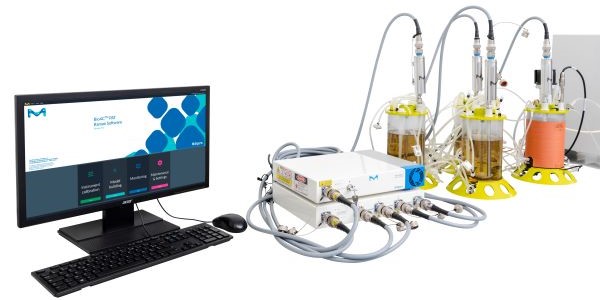
In this webinar, you will discover case studies on the implementation of Raman spectroscopy soft-sensors for monitoring in bench-scale CHO cell intensified seed trains (N-1), steady state, and dynamic perfusion (N) cultures and understand the value of combining automated sampling with Raman spectroscopy


Specifically designed for the bioprocessing industry, ProCellics™ Raman Analyzer with Bio4C® PAT Raman Software enables you to perform in-line and real-time measurement of CPPs and CQAs, from process development to manufacturing
This white paper focuses on PAT and the use of automated sampling technology to accelerate analytical and quality control methods and provide an approach for access to in-line data to monitor processes in real time

In this webinar, you will learn the benefits of automated sampling to the upstream lab, how automated sampling can accelerate access to critical process parameters (CPPs) and critical quality attributes (CQAs) and how automated sampling complements in-line technologies to expand the capabilities for automated measurement of CPPs and CQAs

The MAST® Autosampling Solution is a versatile and modality-agnostic aseptic autosampling solution that ensures the direct collection, delivery, and analysis of samples from multiple sources without manual intervention, providing the user with more time to perform other value-added tasks.