Single-Use Films – Superior Leak Resistance For Challenging, High-Value Bioprocessing Liquid Applications
Single-use technologies can improve efficiency, productivity, reduce capital investment, and minimize cross-contamination in biomanufacturing. For successful implementation, single-use containers made with plastics and polymers must be compatible with bioprocessing requirements such as sterilization by irradiation. However, many bioprocessing films contain an antioxidant, Irgafos® 168, that releases a by-product that adversely impacts growth of Chinese Hamster Ovary (CHO) cells, following gamma irradiation.
Ultimus® film has a fluid contact layer free of Irgafos® 168 and provides the robustness and leak resistance required for large volume bioprocessing applications, such as bioreactors.
Discover more below!
Prevent Costly BaG Leaks in Single-Use Manufacturing

Leaking bags and containers are more than an inconvenience – they directly impact single-use manufacturing costs. Bags made with Ultimus® film deliver superior strength, leak resistance and significant cost savings for multiple fluid management steps particularly those involving transport and shipping of bulk drug substance. Learn more in this infographic.
DownloadDevelopment of an Innovative Single-Use Bioprocessing Film for Strength and Leak Resistance

Films are a key component of single-use technologies used in multiple bioprocessing applications, at both small and large volume scales. This webinar describes how modern analytical testing and new resin selections were leveraged to develop an innovative new film that delivers superior performance for bioprocessing applications.
Register hereSingle-Use Upstream Processing:

Ultimus® film is a single-use, multi-layer film with a fluid contact layer free of Irgafos® 168, a component known to inhibit cell growth. This white paper compares cell culture performance using containers made from Ultimus® film with glass containers. Results confirmed equivalent cell growth, protein production, and protein quality.
DownloadDemonstrated Strength and Durability of Ultimus® Film

The integrity of the film used in single-use systems is critical to maintaining product quality, sterility, and reliable manufacturing operations. This Technical Note summarizes strength and robustness testing for Ultimus® film against ASTM standards. Results highlight its superior performance compared to average performance of commercially available bioprocessing films.
DownloadPerformance of Mobius® Bag Assemblies with Ultimus® Film in Truck Shipping Simulation Test
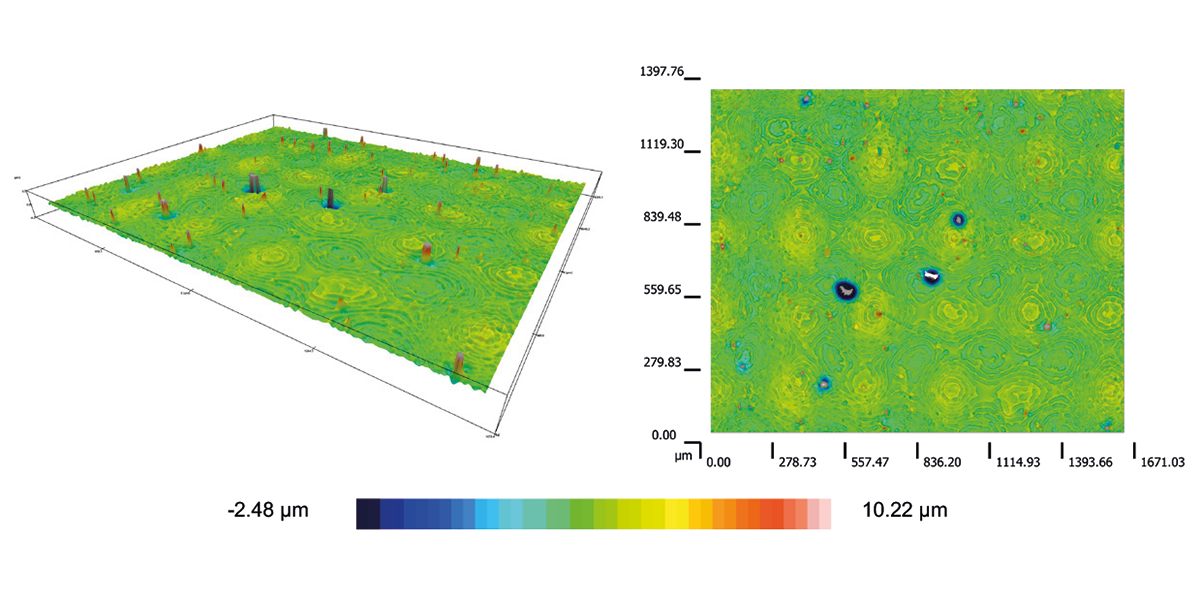
Maintaining container integrity is a key consideration for confidently transporting large volumes of bulk drug substance. This Application Note describes testing with large volume single-use assemblies made with Ultimus® film under representative challenging shipping conditions, resulting in no leak and minimal surface damage despite extensive vibration representing 2400 miles of truck transportation.
DownloadUltimus® Single-Use Process Container Film

Ultimus® film was designed to meet the needs of more challenging single-use applications. This Specification Sheet summarizes key differentiators of this innovative film and the benefits for bioprocessing single-use applications.
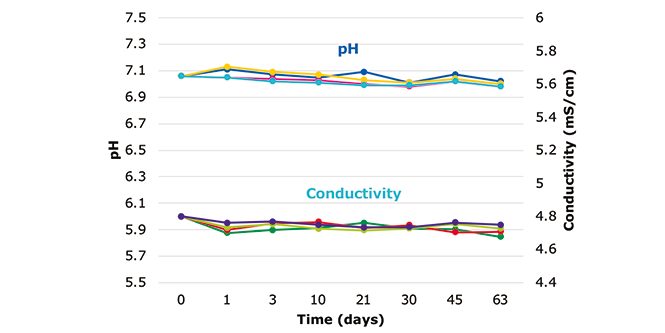
DownloadEnsuring Long Term Stability for Buffers and Cell Culture Media with Single-Use Assemblies Made with Ultimus® Film
The Role of BioPhorum Extractables Data in the Effective Adoption of Single-Use Systems
Before implementing single-use systems in bioprocessing, manufacturers must assess the risk of extractables to patient safety from polymeric components. Read this whitepaper to learn more.
Download