
All Eyes on Biopharma Trends
What are the hot topics in biopharma manufacture? What are the challenges? And where do we need to go? We provide hard data, expert insight, and thoughts for the future in a three part series. Here, we present Part 1.
The Medicine Maker and Ireland’s National Institute for Bioprocessing Research and Training (NIBRT) have collaborated on numerous occasions – perhaps you recall our cover feature from 2016 (here). More recently, The Medicine Maker and NIBRT teamed up to learn more about current manufacturing practices and trends in the biopharma industry, and where the field is heading next, by conducting a survey of industry professionals. You can download the full survey report – for free – here (1).
Our goal? To help inform – dare we say, advance – the biopharma industry by looking at current issues, incoming trends, and exciting innovations. In the next few issues of The Medicine Maker, we’ll be sharing what we’ve learned from the survey, and what some of the esteemed members of our 2018 Power List have to say about the results. In Part 1, we take a look at biopharma therapeutics, now and in the future…
The current state of play
When survey respondents were asked to select the most commercially important biopharma therapeutics available right now, the top answer was monoclonal antibodies (73 percent) – see Figure 1.
“The results align with my own thoughts, as both mAbs and vaccines are growing in significance – especially as the greatest contributors to human longevity continue to be access to clean water, antibiotics, and vaccinations,” says Steve Arlington, President of The Pistoia Alliance. “In the future, their importance to biopharma will depend on the discovery of therapies to treat unmet medical needs, and, in particular, dementias, such as Alzheimer’s, which are becoming more prevalent as our life expectancy increases. We are seeing mAbs, both when used as single therapies or in combination therapies, revolutionize survival rates in many areas of oncology. People also need to realize that mAbs must be used in conjunction with other procedures, such as diagnostics and scanning. The use of mAbs alone will not be enough.”
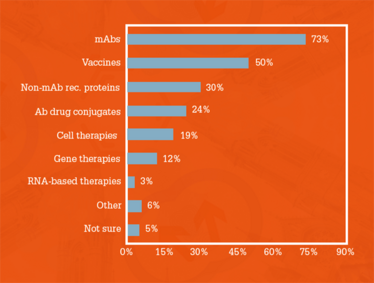
Figure 1. Most commercially important biopharma
therapeutic products currently available.
John Bournas, President and CEO of The International Society for Pharmaceutical Engineering (ISPE), also agreed that mAbs would be important as potential treatments for Alzheimer’s. “mAbs formed the foundation of the industry and will continue to be financially important, even while cost pressures drive firms to seek manufacturing efficiencies,” he says. “Many potential Alzheimer’s treatments are mAbs, and one or more successes in this space would make mAbs production technology and capacity critical for years to come.”
Vaccines were ranked by survey respondents as the second most commercially important biopharma therapeutics. “Vaccines have always been one of the most important class of products for battling infectious diseases – and we should continue to put effort into these to improve quality of life in developing countries. However, their promise for preventing more complicated diseases, such as cancer, is in its infancy, and may not be as broadly applicable as we would hope,” says Marc Bisschops, Director Continuous Bioprocessing at Pall Biotech. “For the immediate future, I definitely agree that mAbs (and mAb derived therapies) will remain the most important class of biopharmaceuticals.”
Therapies of the future
When asking survey respondents what the most commercially important biopharma therapeutics in the next 5–10 years were likely to be, mAbs remained the top answer (56 percent) – as predicted by our experts above – but cell and gene therapies also ranked very high (Figure 2). Bournas says, “Which therapies are most ‘commercially important’ depends on the definition of importance – for the commercial market, mAbs will be important; however, cell and gene therapies will play an important role in the development of new products.”
It’s clear that these cell and gene therapies offer much hype – and so far clinical results are justifying the excitement. However, the Power List members we spoke with, while acknowledging the great potential of these therapies, believe there is still work to be done. In particular, they say it may very well take longer than 10 years for real transformation to be seen.
“The development of cell therapies and gene therapies will move very fast, but personally I think it is unlikely that they will be as important as mAbs in the next 5 to 10 years. It may take a bit longer than that because of the more complicated method of action, as well as manufacturing and administration challenges,” explains Bisschops.
Hal Baseman, Chief Operating Officer of Valsource, also agrees that the cell and gene therapy field may come with teething issues. “In my opinion, advanced therapy medicinal products, such as cell and gene therapies, definitely come with processing challenges. They represent a significant shift from large-scale to small-scale manufacturing methods, and changes in validation, testing, and regulation. Despite this, the field provides opportunities for new companies, facilities, and manufacturing approaches. To what extent this results in a significant increase in product doses remains to be seen.”
Another challenge is commercialization. Arlington says, “At the moment, regardless of whether a breakthrough is on the horizon, pharmaceutical companies can’t work out how to make money from the treatment. Many challenges exist in the process of developing cell and gene therapy treatments, with the additional concern over how a company would deliver and successfully commercialize such a product. The current structures within healthcare providers make it difficult to see attractive delivery models to make these therapies cost effective and commercially viable. We believe this can be overcome, but probably not within the next decade.”
All of this said, such therapies should have a significant impact on human health, in the long term. Bisschops adds, “These therapies could easily become as important as mAbs in the future. Contrary to mAbs, these therapies should – in principle – allow a more sustainable cure for very complicated diseases, such as cancer and auto-immune diseases.”
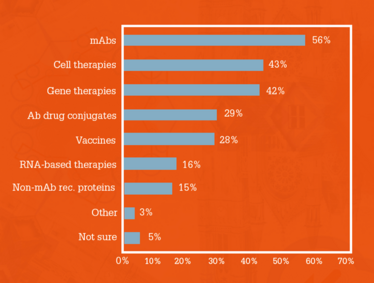
Figure 2. Most commercially important biopharma
therapeutic products in the next 5 to 10 years..
A View From NIBRT
What recent therapeutic innovation has excited NIBRT in the biopharma space?
The FDA approval in 2017 of the first two chimeric antigen receptor (CAR) T-cell therapies: Novartis’ Kymriah and Kite/Gilead’s Yescarta, was certainly a key landmark. But while these therapies provide very promising clinical results, many challenges remain to manufacture such therapies at scale and in a cost-effective manner.
Does NIBRT think mAbs are a hot area at the moment and will they live up to expectations?
Yes, we agree that mAbs will continue to be the most commercially important biologic therapy in the foreseeable future. We’re seeing increasing diversification in the complexity of mAbs, including ADCs, bispecifics, and antibody fragments coming through approvals which require ever more sophisticated manufacturing and supply chain solutions.
What are the institute’s thoughts on the potential of cell and gene therapies? Do you think they’ll be more important than mAbs in the next 5-10 years?
There is certainly very significant potential for these therapies and we’re beginning to see the first investments in commercial scale manufacturing facilities. Despite this progress, there are many scientific, manufacturing and supply chain issues to be addressed if cell and gene therapies are to reach their full potential. We’re really only at the start of this journey.
What issues are most challenging with regard to bringing new biopharma therapies to market?
Key challenges include developing efficient manufacturing and supply chain models that can deliver these therapies to patients in a cost effective manner while maintain the highest levels of quality. This challenge is enhanced by the global shortage of workforces with the required biopharma manufacturing skills and experience.
What do you think industry can do to ease these challenges?
As experts in this article state, collaboration is key. We are beginning to see effective consortia focus on addressing common issues in biopharma manufacturing, although there is a long way to go on this. From a skills perspective, we need to ensure that there is a continued global supply of highly motivated and trained individuals developing their careers in biopharma. Again, there are some excellent initiatives in this area but it requires a constant and ongoing drive.
Ideas and innovations
Cell and gene therapies also stole the spotlight in another part of the survey, when respondents were asked what they considered to be the most exciting therapeutic innovation in recent times (Figure 3). Cell and gene therapy made the top spot by a large margin (57 percent). For the most part, experts from our Power List agree. Bournas says, “Cell and gene therapies are very exciting – the approval of the first product brought us closer to advancing personalized medicine. RNA therapies are another exciting and innovative area of advancement. These therapies have attacked brutal diseases with great success, and that is what makes them so cutting edge.”
“From a therapeutic perspective, personalized medicine and advanced therapy medicinal products are exciting,” says Baseman, “but they and other therapies will also provide the opportunity to develop and advance innovative sterile product manufacturing and process control approaches. From a manufacturing perspective, I’m very interested in continuous process manufacturing and the use of manufacturing intelligence, such as big data, virtual reality, artificial intelligence, robotics and full automation.”
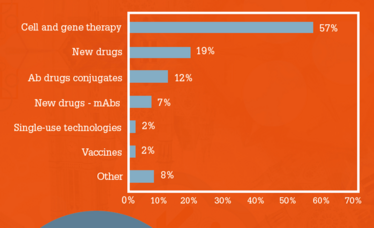
Figure 3. The most exciting therapeutic
innovation in recent times.
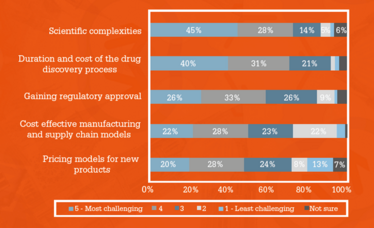
Figure 4. Issues Issues with bringing new biopharma therapies to market: level of challenge.
Other new drugs are also emerging too, and Bisschops says we should not overlook other science. “The first approvals of cell therapy have definitely been exciting,” he agrees. “But I’d also like to add that all of the work on existing biological therapies has resulted in a great accumulation of knowledge and fantastic science that helps us understand complicated diseases and the biology of the human body much better. This should enhance our ability to cure these diseases.”
Arlington, however, adds that true innovation takes time. “Though there is a lot of great science occurring, I would say there haven’t been any innovative breakthroughs related to specific therapies that have excited me recently,” he says. “The innovations that we’re seeing today have been in the works for many years. This is not unusual, and it takes considerable time to reach the proof of concept stage. There are promising leads yet to be confirmed in the oncology field and in non-alcoholic steatohepatitis. Right now, there are certain areas that are gathering speed, such as companion diagnostics, which show great promise and will help to make precision medicine a reality. But despite their potential, the likes of FDA and EMA still need to work out how best to regulate the area before companion diagnostics can be speedily adopted.”
The challenges of scientific progress were also acknowledged by survey respondents. Respondents were asked for their thoughts on the most challenging issues that arise when bringing new biopharma therapies to the market (Figure 4). The issues that respondents felt would be most challenging were the scientific complexities in discovering effective therapies for unmet medical needs and the duration and cost of the drug discovery process. A number of respondents also pointed to the challenges of manufacturing, which Baseman is also concerned about.
“Trying to fit existing manufacturing approaches, regulatory expectations and guidance to the manufacturing methods needed for new therapies is a challenge,” he says. “Traditional approaches to manufacturing, testing and process control may not necessarily align with new therapy manufacturing methods. Trying to force fit these methods may not be the best way to proceed.”
Meanwhile, Bisschops is particularly concerned about the manufacturing hurdles of cell and gene therapies. “For recombinant proteins, vaccines and mAbs, processes are well understood by regulators and biopharma companies, but it is different for newer therapies. Recent successes have demonstrated that there are approaches that work for cell and gene therapies, but it is yet unclear how this will eventually work out. To make it an affordable and scalable process, adequate strategies for manufacturing need to be developed. For instance for cell therapy, the current strategy seems to be to harvest cells from the patient and have them sent to a specialized facility for applying the cell therapy. Once the cells are treated (tested and released), they are sent back to the patient for administration. There may be room for improving the logistics of this treatment, reducing patient risks, inconvenience and costs.”
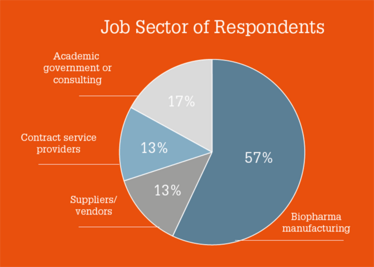
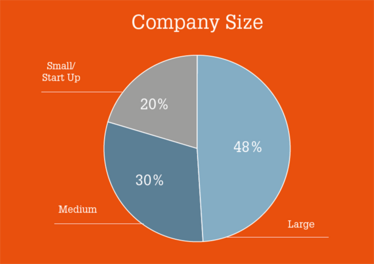
Who Did We Ask?
The survey was fielded from 30 October to 22 November 2017, with over 200 surveys collected. Respondents were based in Europe (45 percent), North America (35 percent), and Asia (16 percent). Four percent came from other regions.
Come together to succeed
Finally, we asked our Power Listers for their advice on what biopharma needs to focus on to rise to the challenges that lay ahead for the field. The key theme is collaboration; the industry must work together to overcome the issues facing the industry.
Arlington: “It is becoming more difficult for individual biopharma companies to come up with answers to industry challenges by themselves. More collaboration is needed at all stages of the R&D process – and regulators and HTAs need to be involved much earlier. Coming together and releasing pre-competitive information will allow the industry to share best practice, enjoy increased efficiency, and reduce costs across the board. As one example, The Pistoia Alliance is involved in a project to increase knowledge of antibody structures – which determine their specific interactions with antigens – in the public domain.”
Baseman: “Scientific and risk-based critical thinking is needed to challenge existing methods and develop innovative and more effective manufacture and process control approaches. A partnership of manufacturers, technology suppliers and regulatory health authorities is the key. Sharing of information between these groups, and across these groups, would reduce the uncertainty and risk of new technologies and approaches. Modernization of manufacturing facilities and processes, and easing of restrictive and burdensome global post-approval change requirements are also important to reduce barriers to innovation.”
A Corking Conference
Want to learn more about the future of biopharma, and hear from international experts within the field?
Inspired by the conclusions from our joint research into industry trends, The Medicine Maker and NIBRT are collaborating on an exciting new conference series focusing on global trends in biopharma and the future of the industry. The inaugural event – Biopharma Trends 2018: Towards Industry 4.0 – will be held on November 13 and 14, 2018, in the Clayton Hotel Silver Springs in Cork, Ireland.
The event will bring together industry leaders to discuss and debate the feasibility of Industry 4.0 and its application to biopharma, covering all the 4.0 building blocks – from cloud computing to big data to smart technology and integrated systems.
Attendees can look forward to unique insights during presentations from renowned presenters, and practical workshops hosted by NIBRT. Speakers include Martin Van Trieste (Parenteral Drug Association), Bernhardt Trout (Novartis and MIT), Jonas Bostrom (EduChem VR and AstraZeneca) and Jim Breen (Janssen).
Industry 4.0 represents the adoption of intelligent, data-driven approaches, and is already bringing tangible benefits to other sectors. But which elements can benefit biopharmaceutical manufacturing, both now and in the future? Is the revolution all empowering – or simply all hype? Join us at Biopharma Trends 2018 to find solutions to the latest issues facing biopharma, and to ask questions of your own. Industry 4.0 is the fourth industrial revolution, and it’s already making its mark on biopharma manufacturing – don’t miss this chance to be part of the conversation!
Bisschops: “I agree; the biopharma community must work together. We all need to realize that we are serving the health of our society – a truly noble goal. A collaborative approach between sponsors, patient organizations, regulatory agencies and suppliers will result in strategies and solutions that eventually provide the most added value to society as a whole – and therefore benefit all partners in the biopharmaceutical community.”
Bournas: “The collaboration of industry and regulatory bodies will pave the way to mastering the new manufacturing paradigm. This is the core of my work at ISPE – the society aims to bring industry and regulators together to work through industry challenges with an open mind to new approaches. Preparing the workforce of the future is also an important initiative for the industry. Workforce availability is a challenge right now – does the industry have the workforce with the right skills to handle the new technologies being developed and the new ways of manufacturing? The Global Pharmaceutical Manufacturing Leadership Forum (GPMLF), in collaboration with ISPE, is taking a close look at industry workforce challenges, and working with industry and academia to identify the skills and training needed by students and young professionals coming into the workforce. They are the future of our industry.”
Indeed, skillsets and training are imperative for the future of the industry – and we’ll be tackling this topic in the second article of this series, which will be published in the August issue of The Medicine Maker.
Meet the Experts

Steve Arlington
President, The Pistoia Alliance
Steve is a Champion of Change on our 2018 Power List because of his work with the Pistoia Alliance. He is passionate about greater collaboration within and between industry, the regulator, the payer, and the provider. Previously, he was Global Lead Partner, Pharmaceuticals and Life Sciences consulting, at PricewaterhouseCoopers.

John Bournas
President and Chief Executive Officer, ISPE
John has been a member of the Power List since 2017. He is responsible for developing ISPE’s global initiatives and business operations, and also leads the organizational efforts in solidifying working relationships with international regulators. Like Steve, he is passionate about collaboration, as well as training the workforce of the future.

Marc Bisschops
Director Continuous Bioprocessing, Pall Biotech
Marc’s work with continuous chromatography and continuous downstream processing earned him a place on the 2018 Power List. He is listed as an inventor on various patents related to continuous process technologies. He hopes to help the biopharma industry become more efficient and agile.

Harold (Hal) Baseman
Chief Operating Officer, ValSource
Hal has over 39 years of experience in the pharma industry and has also been very active in the Parenteral Drug Association, including a stint as Chair. He has been a leader, author, editor and contributor to numerous technical reports, articles, books, and presentations. He is an Industry Influencer on our 2018 Power List.
- NIBRT & The Medicine Maker, “Biopharma Trends: Trends in Biopharma Manufacturing Survey Report” (2017). Available at bit.ly/2lDuHwl.

Making great scientific magazines isn’t just about delivering knowledge and high quality content; it’s also about packaging these in the right words to ensure that someone is truly inspired by a topic. My passion is ensuring that our authors’ expertise is presented as a seamless and enjoyable reading experience, whether in print, in digital or on social media. I’ve spent fourteen years writing and editing features for scientific and manufacturing publications, and in making this content engaging and accessible without sacrificing its scientific integrity. There is nothing better than a magazine with great content that feels great to read.
I have an extensive academic background in the life sciences, having studied forensic biology and human medical genetics in my time at Strathclyde and Glasgow Universities. My research, data presentation and bioinformatics skills plus my ‘wet lab’ experience have been a superb grounding for my role as a Deputy Editor at Texere Publishing. The job allows me to utilize my hard-learned academic skills and experience in my current position within an exciting and contemporary publishing company.



















