Endolysins Vs MRSA
Micreos, a biotech based in the Netherlands, has launched the first endolysin registered for human use against methicillin-resistant Staphylococcus aureus. What is an endolysin? Micreos describes it as a “bacteria-killing enzyme” that originates from bacteriophages. We spoke with Bjorn Herpers from the Regional Public Health Laboratory Kennemerland, Haarlem, in the Netherlands to find out more.
What was the inspiration behind this work?
Antibiotics are one of the major steps forward in medicine, but we now face the consequences of their drawbacks. They not only kill the unwanted bacteria, but the beneficial ones as well, and their use also eventually leads to resistance. Staphefekt was created by scientists from ETH Zurich – Fritz Eichenseher and Martin Loessner – who were inspired by nature’s own solution to overcome these drawbacks. Phages have been recognized as the natural enemy of bacteria, so phage therapy – and in recent years the use of phage endolysins – has been pointed out as a promising new strategy against bacteria.
As a clinical microbiologist, I heard about endolysins in 2006 at the ECCMID congress in Munich, and I was fascinated. In 2012, I read a newspaper article about Micreos, a Dutch biotech company specialized in phage technology. Since I had a freezer full of clinical isolates and was faced with resistance in everyday practice, I decided to get in contact with Micreos to investigate the clinical applicability of their technology.
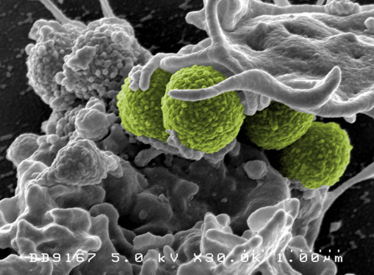
How does Staphefekt work? And does it avoid the issue of drug resistance?
In nature, phages infect the bacterial cell in order to multiply. After new phages have been assembled, the bacterial cell wall is destroyed by phage enzymes called endolysins to allow the newly created phages to be released. In Gram-positive bacteria like Staphylococcus, endolysins work from the outside as well.
Staphefekt is a designer endolysin that selectively kills Staphylococcus aureus, including methicillin-resistant S. aureus (MRSA). It is composed of two parts of naturally occurring phage endolysins: one part is best at specifically binding S. aureus, the other part is best at disrupting its cell wall.
The 3.5 billion years of co-evolution of phages and bacteria has resulted in endolysins that specifically target a certain host species, while other bacteria remain unharmed. Furthermore, the release of the new phages from the bacterial cell is such an essential part of the lifecycle of phages that natural selection has yielded endolysins that effectively target highly conserved structures in the bacterial cell wall that cannot easily be changed. In other words, if bacteria could develop resistance to the current endolysins, they already would have done so.
Tell us about the results from observational patient studies.
We performed two small observational studies. First, we looked at the effect on people carrying S. aureus on their skin. In all three individuals with S. aureus-positive lesions of rosacea, the bacterium was eradicated after applying Staphefekt cream, while other skin inhabitants remained present.
In another series of six patients with S. aureus-related dermatitis (eczema, allergic dermatitis and peri-oral dermatitis), in five of them we saw a decreased burden of S. aureus carriership and diminished symptoms. A couple of them reported less need for corticosteroid use too.
What are the limitations of endolysins?
At this moment Staphefekt is registered in the EU as a medical device for use on intact skin, not for systemic treatment of invasive infections. Endolysins are large molecules that cannot enter tissue cells. Therefore, intracellular infections cannot be treated with these molecules alone. Since endolysins harbor natural epitopes, one could imagine that issues with immunogenicity come into play when administrated systemically. We are currently investigating this.
For local application, for example in burn wounds, surgical wounds, or infected implants, this is much less of a barrier, since Staphefekt works against S. aureus in biofilms. Micreos has also successfully developed an aseptic liquid formulation called Staphefekt XDR.300; one of the technical challenges was to keep the enzyme active throughout the different production steps required for this. As a formulation, XDR.300 is suitable for local administration in research settings. So the next barrier is registration for this use. We are now working on slow-release systems for continuous local treatment; for example near infected implants.
Do you think the dire need for new antibiotics and ways of combating drug resistance will influence more innovative work in the area?
Yes I do - the urgency is felt not only in the medical world, but also by governments and the general public. If this leads to funds becoming available, to faster approval tracks, and to better protection of intellectual property, innovative work will get a boost. There are some good ideas out there. I think that the introduction of the first endolysin for human use has marked the realization of a technological platform for the development of more endolysins and other fields of phage technology, with lots of potential to target other bacteria, like Clostridium difficile.
What are the next steps?
The first steps are to show a beneficial effect in controlled clinical trials. We are collaborating with Professor Suzanne Pasmans, a pediatric dermatologist at the Erasmus Medical Center in Rotterdam who will start an eczema trial in 2015. This kind of evidence is required to convince physicians of its use. The next step would be reimbursement, if Staphefekt is prescribed by physicians as part of their treatment program. As stated above, another goal is approval for local use in wounds; the aseptic liquid formulation needed for this research is already available and we are collaborating with the Association of Dutch Burn Centers on this.
We get a lot of questions from physicians who want to use Staphefekt systemically with their patients suffering from invasive MRSA infections that do not react to therapy. The possibilities for this kind of salvage therapy in a research setting need to be addressed by the physician and the local medical ethical committee, as the product has not been registered for this kind of use. Off course, we want to pursue this in the future.

Making great scientific magazines isn’t just about delivering knowledge and high quality content; it’s also about packaging these in the right words to ensure that someone is truly inspired by a topic. My passion is ensuring that our authors’ expertise is presented as a seamless and enjoyable reading experience, whether in print, in digital or on social media. I’ve spent fourteen years writing and editing features for scientific and manufacturing publications, and in making this content engaging and accessible without sacrificing its scientific integrity. There is nothing better than a magazine with great content that feels great to read.



















