Making the Media of the Future – Today
Biopharma manufacturing continues to advance and the demand for new innovations, such as more efficient cell culture media, is constant.
In previous articles in this series, we heard about intensified perfusion systems from Delia Lyons (1), and about high-density cryopreservation from Jochen Sieck (2). Now, Lyons is joined by Jeremiah Riesberg (Senior Scientist, Perfusion Medium Development, Millipore Sigma, USA) to discuss a novel culture medium. The product, currently in beta testing with chosen clients, has been designed for the most challenging of applications – intensified perfusion at steady state – but is also applicable to a range of other processes.
Why is continued innovation in cell culture media so crucial?
Delia Lyons: Many advances have been made in cell culture media but as the biopharma industry evolves, its needs change – this means we need to continue pushing the boundaries of innovation. Today, clients are demanding faster growing cell lines, higher volumetric productivity, improved product quality, and advanced processes for innovative molecules. In turn, these needs drive demand for improved or entirely novel cell culture media.
We make great efforts to be proactive. Our aim is to anticipate the needs of the market so that we have products ready when the market requires them. The ability to understand the industry before the industry understands itself is essential if you want to be a market leader. However, getting to this position requires a high degree of customer intimacy and close, trust-based client relationships; customers need to feel comfortable sharing future plans and needs, and to know that we listen and consider their input when planning our product development roadmap.
How do available media differ from each other?
DL: In chemical terms, most commercially available media have similar critical components – they all need a particular set of nutrients to meet the basic metabolic demands of cells. One very important distinction between different media is the quality and reliability of the raw materials used in their production. For example, a given nutrient may be available in different chemical forms – and each form may affect cells differently or have lot to lot variability, so it is extremely important to choose the right one. Our raw material characterization team is crucial to ensuring that our products meet the relevant quality standards.
Jeremiah Riesberg: There are many different cell lines, products, and processes in the industry, so the market needs a range of media that can support these different systems. Different biomanufacturing processes, such as fed-batch and perfusion, have unique characteristics, so well balanced medium is desirable to get the best performance from the specific process. It is critical to understand the intended use of the medium, and what needs it must meet.
What is the intended application of your new medium?
JR: Our new catalog medium, EX-CELL® Advanced™ HD Perfusion Medium, has been specifically designed for intensified processes, and tested with steady state perfusion, which is considered very challenging. Maintaining consistent product quality and yield at very high cell densities can be difficult, especially over time periods of 30 or more days. I believe that many companies would welcome a medium that addresses steady state perfusion challenges, and it will be available to them this summer. Furthermore, we expected that a medium that performs well for this most demanding application – maintaining constant productivity over long periods at very low cell specific perfusion rates – would also be applicable to other perfusion-based intensified processes.
How does the new medium compare with the ‘industry standard’?
DL: There is no industry standard! This is the first commercially available medium specifically designed for perfusion processes, so there is no true benchmark yet. We developed it from scratch, using our standard media development tools. We used multivariate statistical analysis to really understand the effect of individual components of the medium. Some components that are beneficial in fed-batch turn out to be excessive in continuous perfusion – more isn’t always better – so to understand which components needed reduction, we had to interrogate individual components of the medium. In other words, we wanted to thoroughly understand the chemistry of the medium and how it affects cells. We tested many different improvements on existing basal media, but the novel medium we developed by optimizing individual components was superior to this. Our product is not just a modified fed-batch medium – and I believe this is a key differentiator.
JR: When we compared our de novo medium to others developed using the traditional approach (adding feed to a basal fed-batch medium), we found the new medium could simultaneously increase the yield by 50 percent while reducing the cell-specific perfusion rate (CSPR) to 40 picolitres per cell per day (pL/cell/day) (Figure 1). With some cell lines, we were even able to maintain a steady state with a CSPR of 20 pL/cell/day.
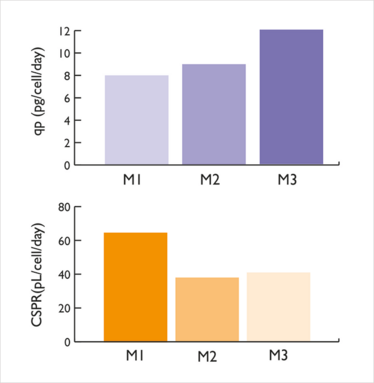
Figure 1: Final medium (M3) could maintain a target cell density with a 40pL/cell/d with a 50 percent increase in specific productivity compared to prototype M1.
What difficulties did you face during development?
DL: It is always difficult to understand and predict current and future market requirements. When you develop an entirely new product, there’s no history to use as a guide – you have to work things out as you go along. We had a number of practical issues to contend with; for example, new media development requires sufficient throughput to enable a reasonable number of conditions to be tested at a small enough scale to remain economically feasible – otherwise you cannot quickly learn and progress.
JR: Exactly. We had to modify our workflow because there was no established small-scale perfusion model or cell separation device with small enough dimensions available. Developing a scaled-down system enabled us to apply the statistical tools that we use to assess the effects of individual components of medium and optimize them specifically for steady state perfusion applications. Developing a small-scale perfusion system in parallel with the development of the medium was a significant challenge. As a team, we all worked very hard on this – and we are very proud of what we have accomplished. I think it is particularly important to note the breadth of applicability. We developed this medium with seven different CHO cell lines producing different types of proteins. From those, four industrially relevant cell lines have been evaluated in steady-state perfusion bioreactors. One of the advantages of Merck KGaA is our in-house cell line development team, who have been invaluable in providing us with the resources to show that the new medium is compatible with a wide range of important cell lines.
How else is Millipore Sigma innovating to meet evolving industry needs?
DL: We appreciate that intensified perfusion applications – steady state or not – are new to the industry, and many clients will benefit from support when they first implement these novel processes. Of course, some companies have been using traditional perfusion processes for labile proteins for years and have a great deal of experience, but many others are relative newcomers. One of our valuable resources is our global team of experts with first-hand experience helping clients set up, optimize, and customize perfusion processes. Today, it is not enough to just develop innovative products – companies should always be looking at how they can add value. We are proud to be part of the teams that offer support and training to customers, helping advance industrial practices and trends.
- D Lyons, “An Intense Focus on Perfusion”, The Medicine Maker, 27 (2017). Available at: bit.ly/2mF5Dqp.
- JB Sieck, “Freezing Down Time in Bioprocessing”, The Medicine Maker, 28 (2017). Available at: bit.ly/2nK7wzD.



















