Taking the mRNA Highway from Development to Commercialization
Driven by the success of COVID-19 vaccines, mRNA products are expanding drug developers’ pipelines. But how can the industry get these formulations to patients more rapidly? It’s important to keep the destination in mind at the very beginning of the journey – and that means paying attention to process robustness, raw materials, and how partners can help.
sponsored by Thermo Fisher Scientific
Perhaps one of the most surprising aspects of the industry’s reaction to the COVID-19 pandemic has been how quickly vaccines were developed. The pandemic has shown what’s possible when the right resources, sufficient government support, and genuine collaboration all come together with a singular focus.
Partnerships have been crucial over the last two years, as it is difficult for developers to move quickly when working alone, particularly in a novel space such as mRNA-based vaccines. Many pharma companies also lacked the supply chains and manufacturing facilities needed to deal with this new modality; both BioNTech and Moderna, for example, relied on partners to help get their COVID-19 vaccines to market. BioNTech partnered with Pfizer – a company with decades of experience in commercializing vaccines – while Moderna leveraged different CDMOs with expertise in manufacturing, formulation, and filling. And both companies partnered with various suppliers to meet the unprecedented demand for raw materials.
mRNA-based COVID-19 vaccines have proven the safety and efficacy of this new modality, resulting in additional drug and vaccine developers exploring the potential of mRNA, and fueling the desire to accelerate development processes to bring these exciting new treatments to patients. Success lies in considering the path to commercialization at the earliest stage possible. Here, process and raw material characterization are necessities as is an understanding of regulatory requirements. Choosing the right partners for the journey can also make a huge difference.
“The process is the product” no longer
With traditional viral vaccines, the end product is a complex biologic, and there is often limited analytical characterization. With these types of products, a common mantra is, “the process is the product.” This mindset refers to the fact that it is difficult to fully understand the impact that process, scale, and raw material changes may have on the end product’s safety and efficacy. Because of these challenges, vaccine development is slow, with production typically “scaled out” rather than “scaled up” to minimize process changes. For example, a traditional vaccine developer might scale out by moving from a single roller bottle to tens of thousands of roller bottles.
mRNA processes are different. With mRNA, it is possible to characterize both the end product and the materials used to manufacture it, enabling a true quality by design (QbD) approach and the ability to “scale up” from a 1 mL to a 50 L volume while mitigating risk to the end product.
A QbD approach identifies and characterizes critical process parameters (CPPs) and critical quality attributes (CQAs) so manufacturers can demonstrate both process and end product consistency to regulators. Indeed, keeping this goal in mind early on should accelerate process development by eliminating batch failures and potential rework at later stages. This approach allows for data-driven justification and understanding during process changes or fluctuations – “the process is the product” no longer.
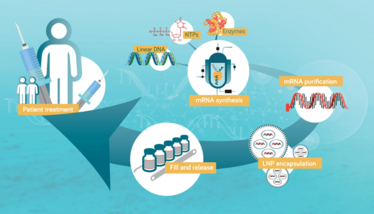
The typical process for mRNA synthesis begins with plasmid DNA production. The linearized DNA feeds into the in vitro transcription (IVT) reaction, which adds nucleoside triphosphates (NTPs) and enzymes to convert DNA to mRNA. From there, the mRNA must be purified to remove any impurities resulting from the process, including residual DNA. Then, it’s onto lipid nanoparticle (LNP) encapsulation and the final formulation and fill before the final product is released and made available for distribution and patient administration.
From a process perspective, there are potential challenges and risks in each of these steps. To start, it’s important to get the right balance of NTPs, enzymes, and DNA to optimize yields. When moving to purification, all steps need to be optimized to reduce product loss while maximizing the purity of the resulting single-stranded capped mRNA. As the industry gains more experience with mRNA, we expect these parts of the manufacturing process to improve, driving more robust, improved process efficiencies.
The encapsulation portion of the process will also evolve as the complexity of mRNA drugs increases. Current COVID-19 vaccines encode a single protein – the spike protein of SARS-CoV-2 – and encapsulate the mRNA in LNPs. Other mRNA products coming through development pipelines are significantly more complex, with LNPs encapsulating five or six different mRNA types encoding multiple antigens. These components all need to be combined in the correct ratio and in a consistent manner. Moving forward, optimizing this part of the process could present one of the biggest mRNA production challenges.
High-quality products are built with high-quality raw materials
Careful consideration of the quality of raw materials is crucial to mRNA process robustness. Controlling raw materials helps to control the process and, ultimately, the end product. For example, a specific enzyme may impact the reaction kinetics of the mRNA process (a potential CPP), which could lead to impurities, such as dsRNA (a CQA), that ultimately ultimately affect how the drug works in the body. Additionally, the impurities associated with raw materials have the potential to impact CQAs directly if there is a risk of introducing adventitious agents into the process.
Once the CPPs, CQAs, and their relationships to raw materials are understood, a drug developer can establish raw material requirements and then work with suppliers to ensure the products they receive meet those activity and purity specifications. For example, enzymes, which are primarily produced through a fermentation process, can potentially introduce adventitious agents, depending on how they are manufactured. Drug developers can mitigate this risk by selecting enzymes produced with animal origin–free components and using single-use fermentors.
If you’re developing an mRNA vaccine, your worst-case scenario would be buying a low-cost enzyme, using it for scale-up, and then learning it is manufactured with bovine serum albumin (BSA). You would then need to either identify another source for the enzyme that doesn’t use BSA and rework your process with the new enzyme or perform a detailed risk assessment to demonstrate that the original enzyme doesn’t introduce a risk of transmissible spongiform encephalopathy (TSE) or bovine spongiform encephalopathy (BSE). You would also need to continually track and manage risk associated with each lot of enzyme.
Support across the mRNA workflow
From small start-up biotechs to global biopharma companies, we continue to see industry investments in mRNA. mRNA technology and manufacturing processes are still immature relative to other modalities, but mRNA vaccines and therapeutics show incredible promise in fulfilling currently unmet medical needs. Over the next decade, we expect continued advances in the development of mRNA-based vaccines and therapeutics.
Because of the specific challenges associated with manufacturing mRNA, drug developers will almost certainly continue to turn to partners with complementary experience and skills. The utility of this approach was demonstrated by BioNTech and Moderna, as they were ultimately successful by maintaining focus on their respective technologies while allowing partners to do the heavy lifting in commercialization and manufacturing. Indeed, the most fruitful partnerships will be those that allow each player to concentrate on their own core competencies to drive success and overcome challenges, including enzyme engineering, chemical synthesis, drug development, and GMP manufacturing.
I think we can all agree that mRNA is exciting. But it is also new, so regulatory requirements are still evolving. Successful companies will continue to rely on the best practices, such as QbD, that were developed for other biologics while keeping in mind that regulatory requirements may change. To be prepared for that evolution, companies should gain a very firm grasp of their processes and raw materials. In this regard, good partners love to help. At Thermo Fisher Scientific, for example, we’ve already collaborated with customers across the mRNA workflow, from discovery to commercialization.
The COVID-19 pandemic has highlighted the many benefits of collaboration and partnership in delivering a new vaccine technology to meet critical demand. Now, we need to use the lessons learned from this unprecedented challenge to bring new therapeutics to other patients in need just as quickly and safely.
Serena Smith, Director of Market Intelligence & Strategy – mRNA Vaccines & Therapeutics at Thermo Fisher Scientific



















